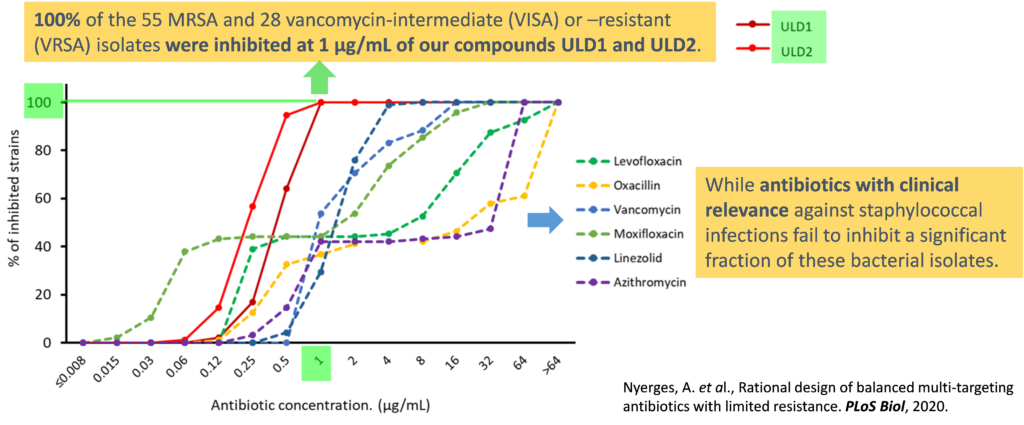
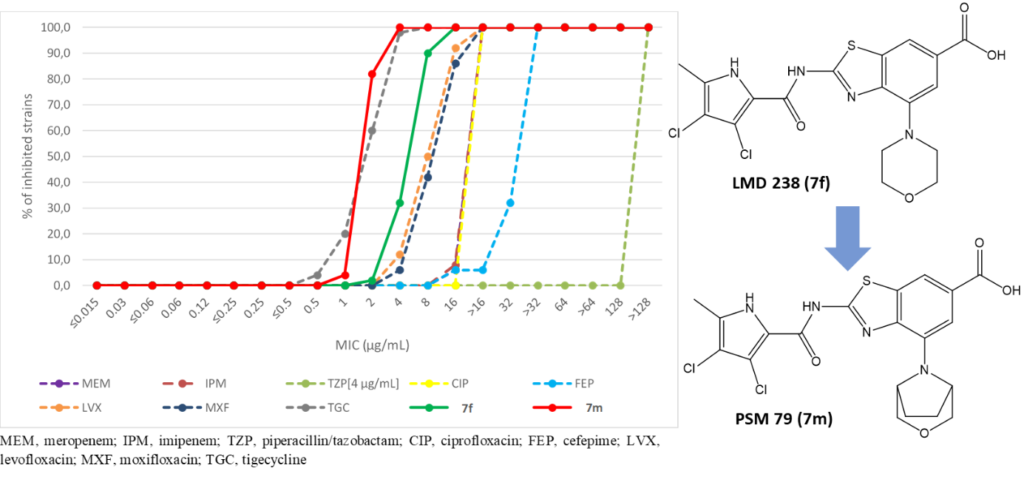
One promising strategy to address the rapid evolution of bacterial resistance is the design of antimicrobial compounds that equipotently inhibit two bacterial targets. The rationale for this approach is that the development of resistance to multitargeting antibiotics (MTA) would require the simultaneous occurrence of multiple specific mutations at both targets, which is extremely rare. Therefore, multitargeting antibiotics should be less susceptible to resistance compared to monotargeting antibiotics.
The core focus of our research revolves around addressing the urgent need for effective antibacterial agents against ESKAPE pathogens. Our target product profile includes combating methicillin-resistant (MRSA), vancomycin-intermediate (VISA) Staphylococcus aureus, clinical isolates of Acinetobacter baumannii, and mycobacteria. To achieve this, we are working towards the development of inhibitors that demonstrate a balanced potency against bacterial DNA gyrase and topoisomerase IV. Specifically, we are targeting amino acid residues that are less susceptible to spontaneous mutations, as any mutations in these residues would impair the functionality of the target enzymes. Over the years, we have compiled an extensive library of potent inhibitors that effectively target both DNA gyrase subunit B and topoisomerase IV subunit E. These inhibitors exhibit a broad spectrum of antibacterial activity, proving effective against multidrug-resistant pathogens and demonstrating efficacy and safety in mouse infection models.