Ion channels are now considered unconventional, promising oncological targets, whose expression is often altered in cancer cells and which are emerging as critical players in tumorigenesis. The idea of targeting Kv1.3 ion channels directly in mitochondria, whose function critically depends on ion fluxes and which are crucial for both cell survival and apoptosis, could change the therapeutic field of cancer research. Resistance to apoptosis is one of the key hallmarks of cancer cells and often arises as a mechanism to escape drug-induced toxicity. Kv1.3 is also important for immune cells, which are a central component of the tumour microenvironment, both at the primary site and–more importantly for metastasis–at the distant location of the metastatic tumour. Therefore, it is reasonable to postulate that ion channel-based therapies may be beneficial in preventing and eradicating metastasis and may be useful in cells that are resistant to classical chemotherapy.
Chemical Strategies and Solutions for Bacteria and Cancer cell Resistance
ARIS J7-4635: MitoCan – Preclinical development of new Mitochondrial ion channel inhibitors for Cancer therapy; 2022-2025; (1.5 million)

Presentation of MitoCan targeting mitochondrial ion channels Kv1.3 (mitoKv1.3) in cancer. ROS: reactive oxygen species; PK: pharmacokinetics; PADMET: physicochemical properties, absorption, distribution, metabolism, elimination, and toxicity; PDAC: pancreatic ductal adenocarcinoma.
MitoCan is an innovative project aimed at targeting cancer by utilizing mitochondrial Kv1.3 ion channels with proof of principle in in vivo model of pancreatic ductal adenocarcinoma (PDAC). This is very relevant because the incidence of PDAC, the third leading cause of cancer-related mortality, is expected to rise. Therapeutic options for patients with metastatic disease offer only modest survival benefit as PDAC is recalcitrant to both conventional and immune-based therapies.
MitoCan is based on a patent application (MITOCHONDRIOTROPIC BENZAMIDE POTASSIUM CHANNEL KV1.3 INHIBITORS) and newly developed results, which indicate that MitoCan selective and potent mitochondrial Kv1.3 inhibitors have a great potential for lead optimization and preclinical development. Mitocan joins international partners that are the discoverers and undisputed leaders in research of cancer and ion channels in cancer together with the most important Slovenian research institutes to make a breakthrough in the potential of ion channels for the treatment of cancer.

MitoCan multidisciplinary Consortium in anticancer drug discovery of novel mitochondrial ion channel inhibitors.
ARIS J1-3030: MTAvsAMR – new MultiTargeting Antibiotics against AntiMicrobial Resistance; 2021-2024; 300k EUR
There is an urgent need for new therapies and new antibiotics to treat deadly infections caused by so-called ESKAPE pathogens – bacteria that are often resistant to available antibiotics. Not only infections caused by these pathogens are difficult to treat, but discovering new therapies to overcome Gram-negative resistance is particularly challenging. The World Health Organization classifies these pathogens as a critical threat because they are resistant to almost all available antibiotics. More than 95% of antibiotics in clinical trials are being developed by small companies and academia, not the big pharmaceutical companies that once dominated the field.
Ensuring that the supply of new antibiotics keeps pace with these evolving pathogens will require a robust pipeline of new antibiotics and innovative solutions. One promising strategy to address this rapid evolution of resistance is the design of antimicrobial compounds that equipotently inhibit two bacterial targets. The rationale for this approach is that the development of resistance to multitargeting antibiotics (MTA) would require the simultaneous occurrence of multiple specific mutations at both targets, which is extremely rare. Therefore, multitargeting antibiotics should be less susceptible to resistance compared to monotargeting antibiotics. However, the design of antibiotics that equipotently inhibit two bacterial targets and exhibit potent antibacterial activity, low frequency of resistance, adequate safety and physicochemical properties remains a challenge.
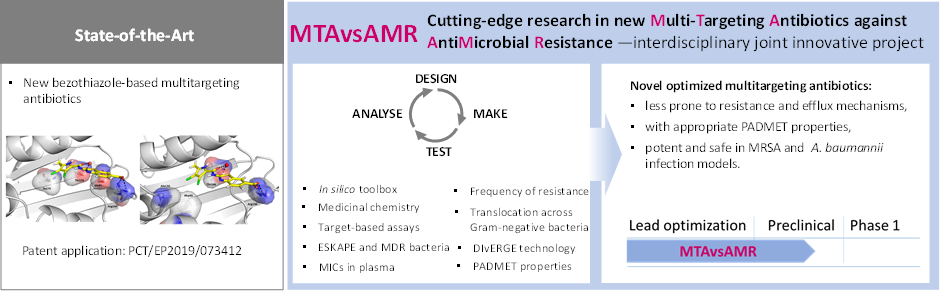
Presentation of the MTAvsAMR antibacterial drug discovery project. MDR: Multi Drug Resistant; MIC: Minimal Inhibitory Concentration; DIvERGE: Directed Evolution with Randomized Genomic Mutations; PADMET: Physicochemical properties, Absorption, Distribution, Metabolism, Elimination and Toxicity.
In the MTAvsAMR research project, we aim to develop a new structural class of MTA against two well-established molecular targets with innovative approaches to potent and safe antibiotics with limited resistance. Our MTAs are expected to simultaneously meet the following criteria: First, a balanced potency against the two essential bacterial targets DNA gyrase subunit B and topoisomerase IV subunit E is required. Second, they should establish strong intermolecular interactions at multiple, functionally important amino acid positions within the binding sites of their target proteins. Such interactions are thought to make spontaneous resistance unlikely, as mutations at these sites would impair the functionalities of the target proteins. In addition, we will target the antibacterial activity of the new molecules against ESKAPE pathogens to address an unmet medical need, with a target product profile of methicillin-resistant (MRSA), vancomycin-intermediate (VISA) Staphylococcus aureus and Acinetobacter baumannii clinical isolates.
ARIS J1-3031: Development of new inhibitors of bacterial topoisomerases to overcome antimicrobial resistance; 2021-2024; 300k EUR
The ATP binding site on DNA gyrase and topoisomerase IV is an attractive target for the development of new antibacterial agents. DNA gyrase and topoisomerase IV share 40% sequence identity, providing an exceptional opportunity for dual targeting. In recent years, several small molecule classes of ATP-competitive inhibitors have been discovered, but few have reached the market.
The overall aim of the project is to discover new ATP-competitive small-molecule inhibitors of DNA gyrase and topoisomerase IV with activity against resistant bacterial strains. We will start from our recently discovered N-phenylpyrrolamide GyrB/ParE inhibitors and optimise them using innovative structure-based design. We will design original new compounds with balanced dual-targeting activity towards GyrB and ParE, by interacting with multiple, functionally essential amino acids of both target proteins, with low probability of resistance development. We will determine the inhibitory activities of the prepared compounds on bacterial topoisomerases and on various off-targets. Potent and selective inhibitors will be subjected to detailed microbiological evaluation on selected Gram-positive and Gram-negative bacteria and mycobacteria, including multidrug resistant (MDR) strains. For the most promising hits with potent antibacterial activity, a comprehensive medicinal chemistry programme will be pursued to develop them towards lead compounds, which will involve determination and optimisation of compounds’ physicochemical (logP, logD, thermodynamic and kinetic solubility) and ADMET properties. The result of the project will be new lead compounds with potential to be developed into clinical candidates for the treatment of infections caused by resistant bacterial strains.

Representative N-phenylpyrrolamide GyrB/ParE inhibitor A and an overview of the work programme.
FINISHED GRANTS
University of Ljubljana Innovation Fund: Fight against bacterial resistance: new antibacterial agents against resistant bacteria, PI: prof. dr. Lucija Peterlin Mašič (2020-2021) (19k EUR)
University of Ljubljana Development Fund: Synthesis of the Week and Synthetic Masterchef of the Faculty of Pharmacy, PI: assist. prof. dr. Andrej Emanuel Cotman (2022–2023) (12k EUR)
ARRS project J1-1717: Development of novel Hsp90 inhibitors with anticancer activity, PI prof. dr. Tihomir Tomašič (2019-2023) (300k EUR)
ARIS project Z1-2635: Modular asymmetric total synthesis of bioactive multi-chiral natural products, PI: assist. prof. dr. Andrej Emanuel Cotman (2020–2022) (100k EUR)
ARRS project J1-9192: New antitumor agents against potassium ion channels hEAG1 and their validation in lymphomas, PI: prof. dr. Lucija Peterlin Mašič (2018-2021) (300k EUR)
FWO project: Discovery and mechanism of action of novel hEAG1 potassium channel lead molecules with anti-cancer activity, PI: prof. dr. Lucija Peterlin Mašič (2019-2022). Acronym: DISCO-a-GOGO (270k EUR)
IMI ENABLE: European Gram-negative Antibacterial Engine; hit-to-lead optimisation of new dual DNA gyrase B and topoisomerase IV inhibitors, PI: prof. dr. Danijel Kikelj, Chemistry leads: prof. dr. Lucija Peterlin Mašič, prof. dr. Tihomir Tomašič (2019-2020) (500k EUR)
CELSA project: Collaborative research project UL Ljubljana, KU Leuven: Discovery of New Leads of Voltage Gated Potassium Ion channels as Emerging Cancer Targets, PI: prof. dr. Lucija Peterlin Mašič (2017-2020), (30k EUR)

